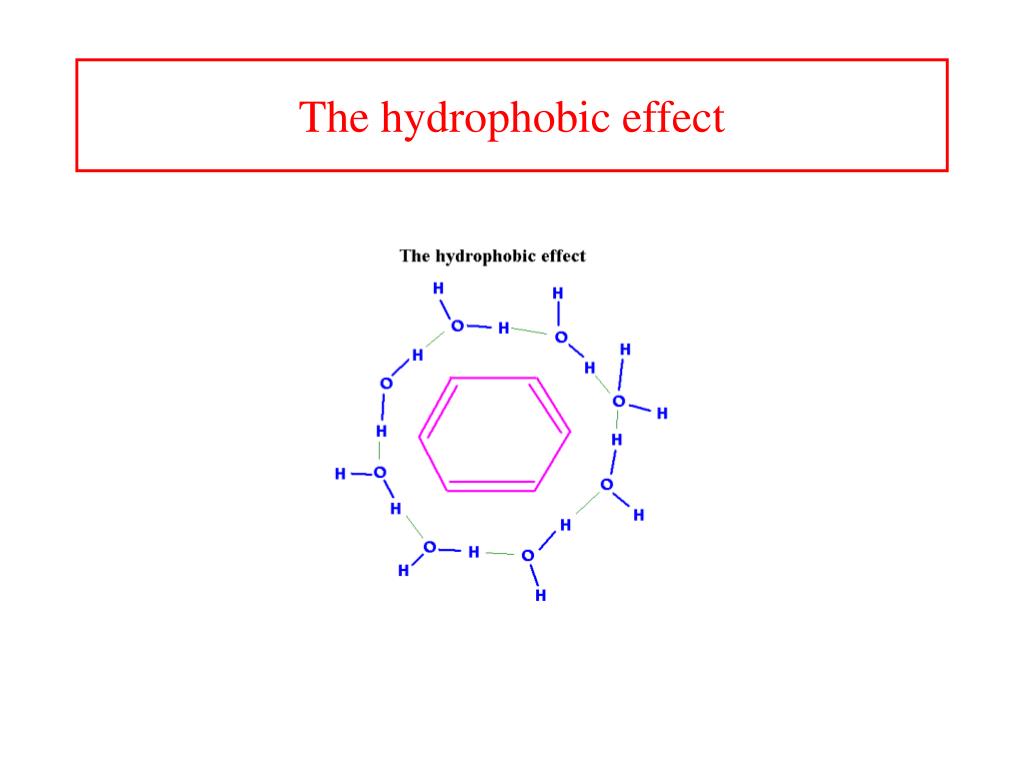
   The folding transition abruptness and van't Hoff-to-calorimetric-enthalpy ratio are found to increase dramatically by this simple and physically motivated mechanism. Thermodynamics are governed by the hydrophobic energy function, according to which hydrophobic monomers tend to make contacts unspecifically while the reverse is true for hydrophilic monomers, with the additional restriction that only contacts between monomers adopting one of zh  In addition to the square lattice, with z=3 conformations per monomer, we use extensions in which diagonal step vectors are allowed, resulting in z=5 and z=7. This scheme is intended to mimic the requirement that polar backbone groups of real proteins must form hydrogen bonds concomitantly to their burial inside the apolar protein core. Conformational restrictions imposed by hydrogen bond formation during protein folding are investigated by Monte Carlo simulations of a non-native-centric, two-dimensional, hydrophobic model in which the formation of favorable contacts is coupled to an effective reduction in lattice coordination. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
